Unit 3, B.3 #1-4, page 344
1) Why is human exposure to ultraviolet radiation potentially more harmful than exposure to infrared radiation?
Because infrared radiation is essentially heat energy, exposure to it is not as harmful as exposure to ultraviolet light, which is the most energetic form of sun radiation. Ultraviolet light can cause sunburns and skin cancer and can even sterilize materials by killing bacteria and destroying viruses (UV-C radiation). UV-C radiation can damage living organisms due to its energy that is high enough to break covalent bonds.
2) Describe two essential roles played by visible solar radiation.
Visible solar radiation energizes electrons in some chemical bonds. For example, this provides the energy needed for photosynthesis reactions and the visible scattering of light during sunset.
3) Explain why dry, arid regions in the United States, such as New Mexico and Arizona, experience wider air-temperature fluctuations from night to day than do states with more humid conditions, such as Florida.
Since there is less water vapor, and therefore, less greenhouse gas caused by water vapor in dry and arid regions such as New Mexico and Arizona, infrared radiation is not as stored and reradiated and reflected back into the atmosphere from the entering of UV and visible light that is transformed into IR radiation in clouds, but the visible and UV radiation directly exposed to the surface of the earth (bare in mind that most never reaches earth's surface) is used to heat earth. This results in wider air-temperature fluctuations from night to day than states with more humid conditions, for not as much heat is stored in the clouds.
4) Suppose Earth had a less dense atmosphere (fewer gas molecules) than it does now:
a. How would average daytime temperatures be affected? Why?
b. How would average nighttime temperatures be affected? Why?
a. The daytime temperatures would be much cooler, because with less greenhouse gases such as CO2 and H2O (g), less infrared radiation would be stored and reradiated and reflected back into the atmosphere from the transformation of UV and visible radiation to IR radiation in clouds (since actual IR radiation from the sun is reflected back into space). Less stored heat energy would enter throughout the day.
b. Without stored heat energy in the atmosphere, nights would be very cold without the presence of the sun!
4SBS #14-20, page 361
14) Describe how atmospheric CO2 and water vapor help maintain moderate temperatures at Earth's surface.
CO2 and water vapor are both greenhouse gases, atmospheric gas molecules that effectively absorb infrared radiation-- this infrared radiation is not directly from the sun, but is the product of transformed UV and visible radiation from the sun within the clouds. Infrared radiation is essentially heat energy, that these greenhouse gases have made from trapping UV and visible light in order to absorb, reradiate, and reflect the heat energy onto earth's surface.
15) List two natural processes and two human activities that can increase the amount of
a. CO2 in the atmosphere.
b. CH4 in the atmosphere.
a.
Natural: 1. breathing; 2. bacterial decay.
Human: 1. agricultural and industrial activities; 2. burning of fossil fuels.
b.
Natural: 1. decomposition of plants; 2. decomposition of animal wastes
Human: 1. Refining fossil fuels; 2. raising of livestock.
16) What changes in the composition of the atmosphere would cause the average surface temperature of the Earth to
a. increase?
b. decrease
a. lower altitude, increase in greenhouse/atmospheric gases, higher air pressure.
b. higher altitude, decrease in greenhouse/atmospheric gases, lower air pressure.
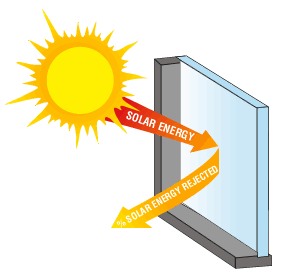
17) Explain why, on a sunny winter day, a greenhouse with transparent glass walls is much warmer than is a structure with opaque wooden walls.
What goes on in greenhouse with transparent glass imitates the way the greenhouse gases transform UV and visible radiation to IR radiation within clouds, and reradiate and reflect the heat energy within earth, or the greenhouse (since the infrared light cannot escape through the glass, like earth's CO2 and H2O gas that act as a shield). This is why on a sunny winter day, a greenhouse with transparent glass walls is much warmer than is a structure with opaque wooden walls.
18) Draw sketches to show how
a. a greenhouse works.
b. the global greenhouse effect works.
a.
b.
19) List three chemical reservoirs of carbon atoms.
Four chemical reservoirs of carbon atoms include atmospheric CO2 gas, solid calcium carbonate (CaCO3) in limestone, natural gas (methane CH4), and organic molecules.
20) Explain how, over time, a particular carbon atom can be part of the atmosphere, biosphere, lithosphere, and hydrosphere.
Carbon naturally moves within Earth's systems. The carbon atom could be part of the atmosphere as a gas as a result of photosynthesis, in the lithosphere as plant or animal waste decay, or part of the hydrosphere as limestone.




A forex trading platform is a powerful digital tool that enables traders to buy and sell currencies in the global foreign exchange market with speed and precision. It provides real-time price quotes, advanced charting tools, technical indicators, and order execution features that help traders analyze market trends and make informed decisions. Whether you are a beginner learning the basics or an experienced trader applying complex strategies, a reliable forex trading platform offers the performance, security, and flexibility needed to trade confidently across multiple currency pairs and market conditions.
ReplyDeleteContact us Address – 1st Floor, The Sotheby Building, Rodney Bay, Gros-Islet, SAINT Lucia P.O Box 838, Castries, Saint Lucia Phone no – +97144471894 Website – https://winprofx.com/